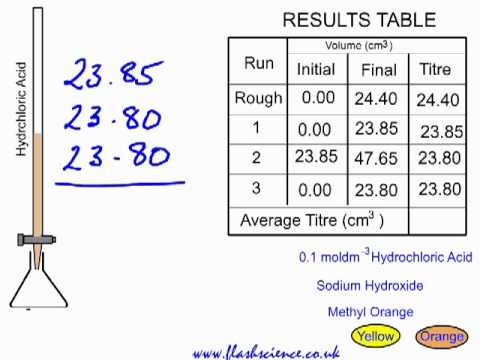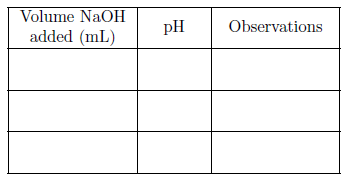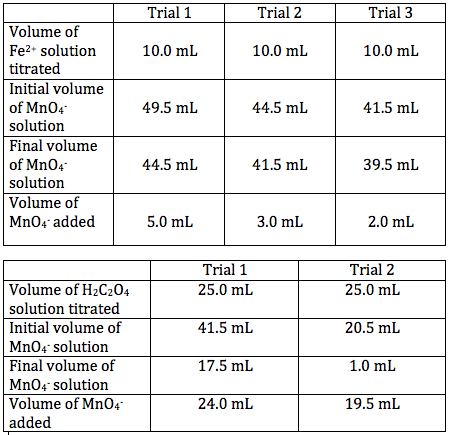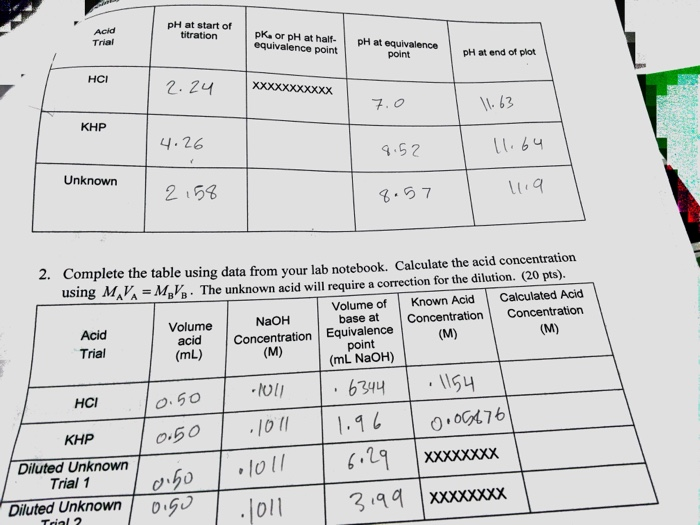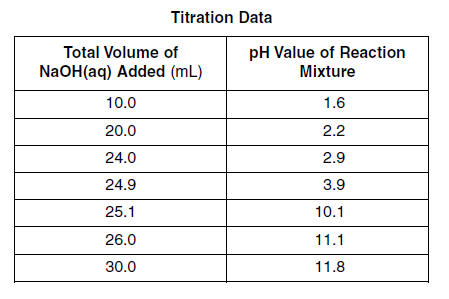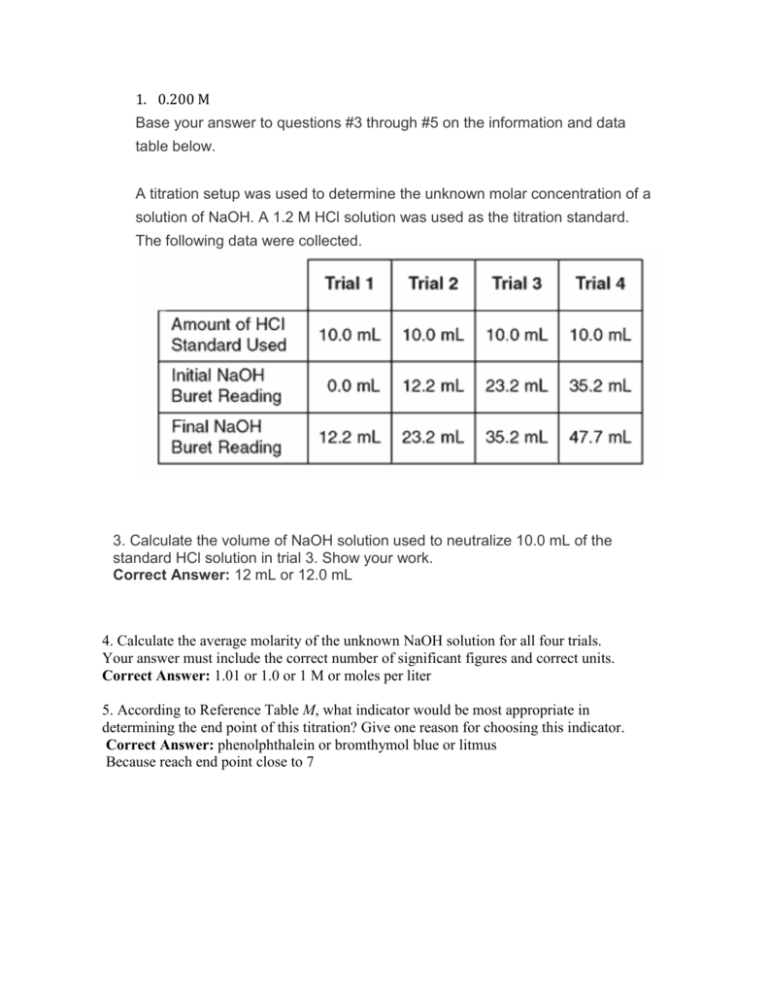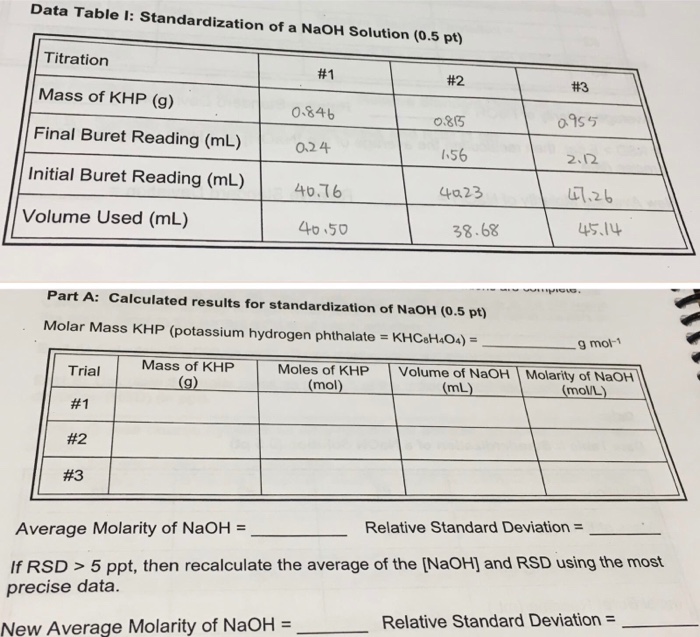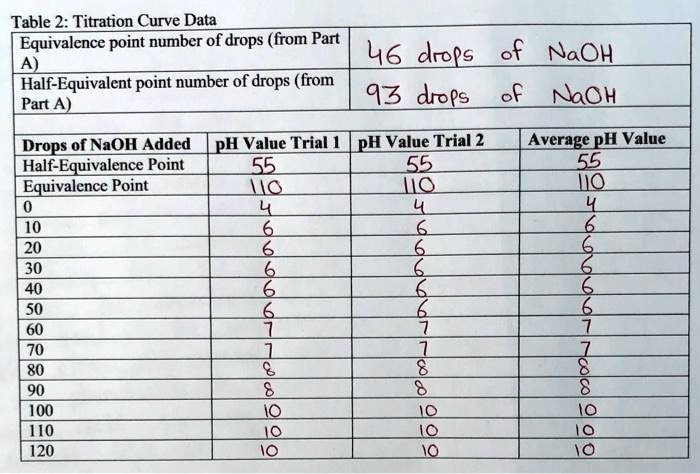
SOLVED: Table 2: Titration Curve Data Equivalence point number of drops (from Part 46 drels of NaOH A) Half-Equivalent point number of drops (from 93 drols of Part A) Noh Drops of
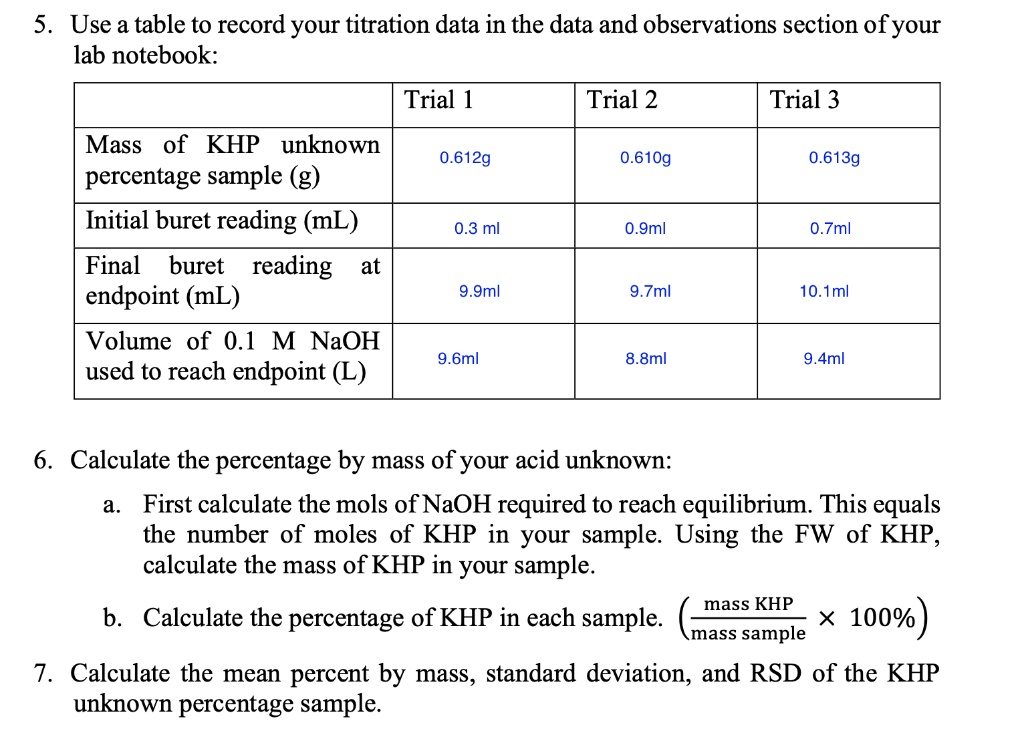
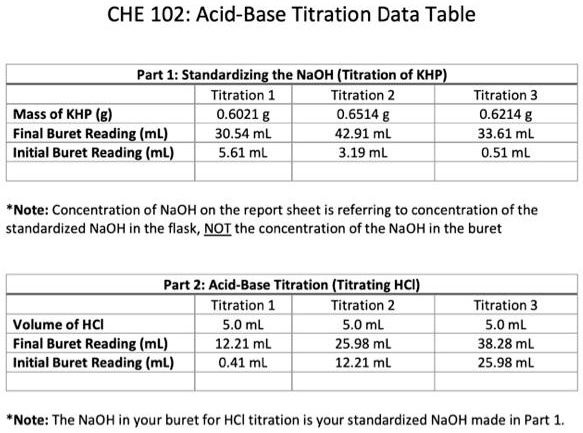
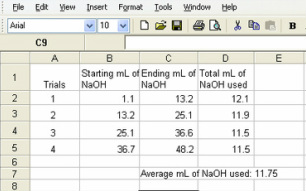
SOLVED: 5 Use table to record your titration data in the data and observations section of your lab notebook: Trial Trial 2 Trial 3 Mass of KHP unknown percentage sample (g, Initial
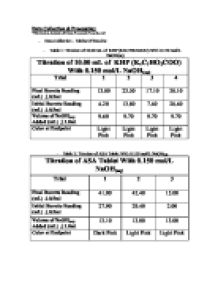
The purpose of this Titration Analysis of ASA experiment was to use titration analysis techniques to determine the amount of ASA content contained of a standard pain-relief tablet. - International Baccalaureate Chemistry -